Introduction
Chemical reactions are at the heart of countless industrial processes — but when they behave unexpectedly, the consequences can range from process upsets to catastrophic incidents. Understanding how a material responds to heat, pressure, or mixing with other substances is essential before that material ever reaches full-scale production.
Reactive chemical testing provides that understanding. Through controlled laboratory analysis, engineers can identify thermal instability, decomposition risks, and runaway potential before they become real-world problems. This article walks through the primary testing methods used to assess chemical reaction hazards — what each technique measures, what data it produces, and how that data supports safer process design.
Differential Scanning Calorimetry (DSC)
DSC is typically the first test performed in a reactive chemical hazard assessment. It measures heat flow during controlled heating, detecting exothermic and endothermic events that indicate thermal instability, decomposition, or phase transitions. The applications of DSC in process safety are wide-ranging — it can be used to screen for thermal hazards, assess chemical incompatibilities, characterize materials, and evaluate the thermal stability of products. Because it requires only a small sample (typically 5–20 mg) and produces results quickly, DSC is well-suited as a screening tool before committing to more resource-intensive testing.
The sample and an inert reference are subjected to the same temperature program. Any difference in heat flow between the two reveals significant thermal events within the sample. For safety assessments, sealed high-pressure cells are recommended to prevent evaporative losses. Tests are typically run at a ramp rate of 1 to 10 K·min⁻¹ — higher ramp rates reduce sensitivity in determining onset temperature. Running tests in both air and nitrogen atmospheres helps distinguish oxidative reactions from pure decomposition.
A key safety threshold to note: decomposition energy exceeding 800 J·g⁻¹ indicates potential explosive properties. The onset temperature obtained from DSC should be used with an appropriate safety factor, as it is influenced by the phi factor and heat losses during testing. For more precise onset temperatures, adiabatic calorimetry is recommended as a follow-up.
From DSC testing, you can obtain:
- Reaction heat (J·g⁻¹)
- Onset temperature of exothermic or endothermic reactions
- Reaction kinetic parameters (activation energy, reaction order, pre-exponential factor) — requires multiple tests at variable scanning rates
- Induction time for autocatalytic reactions
The applicable testing standard is ASTM E537 — Standard Test Method for the Thermal Stability of Chemicals by DSC.
Differential Thermal Analysis (DTA)
DTA evaluates thermal behavior by monitoring the temperature difference between a test sample and an inert reference material during controlled heating. When the sample undergoes a thermal event — melting, crystallization, decomposition, or gas evolution — its temperature deviates from the reference, revealing the nature and intensity of that event. Common equipment used for DTA testing includes the Carius tube and the Rapid Screening Device (RSD), both of which allow simultaneous pressure monitoring, which is critical for detecting gas evolution or runaway reactions.
While DTA is semi-quantitative compared to DSC, it is highly effective for screening reactive or unstable substances and is particularly valuable for detecting exothermic decomposition and phase instability early. It is often used alongside other screening methods to build a more complete picture of a material’s thermal behavior.
From DTA testing, you can obtain:
- Onset temperatures (with appropriate safety margins)
- Qualitative reaction intensity
- Vapor-liquid equilibrium data
- Total gas output
Though no single universal standard governs all DTA applications, the method aligns with general thermal analysis practices referenced in ASTM E537 and company-specific safety protocols.
Thermogravimetric Analysis (TGA)
TGA measures changes in a sample’s mass during controlled heating, conducted in either an inert or reactive atmosphere. By tracking exactly how much mass is lost and at what temperature, TGA reveals a material’s composition, thermal stability, and decomposition behavior across a range of process conditions. Common applications include measuring moisture content, detecting volatile components, analyzing decomposition patterns, and assessing oxidative stability.
In process safety, TGA is particularly valuable for identifying safe operating temperature ranges and detecting the onset of decomposition. It can also reveal incompatibilities between materials and process conditions — for example, identifying temperatures at which flammable or toxic gases begin to evolve. The first derivative of the TGA curve (the DTG curve) helps pinpoint inflection points for more detailed interpretation.
From TGA testing, you can obtain:
- Mass change versus temperature
- Mass change versus time
- Reactivity in air, oxygen, or other reactive atmospheres
- Decomposition or reaction kinetics
Relevant ASTM standards include E1131 (composition analysis), E1641 (decomposition kinetics), E2550 (thermal stability), and E1868 (loss-on-drying).
Accelerating Rate Calorimetry (ARC)
ARC is used to evaluate the thermal stability and runaway potential of a substance or mixture under adiabatic conditions — meaning no heat escapes the system, simulating a worst-case industrial scenario. It is particularly valuable for detecting slow self-heating reactions that might not be apparent in faster screening tests like DSC, and for substances with autocatalytic behavior or delayed exotherms.
The instrument operates primarily in heat-wait-search (HWS) mode. The sample is heated step by step, and after each increment the system waits for thermal equilibration before searching for any self-heating above the detection threshold (typically 0.02 K·min⁻¹). Once exothermic activity is detected, the instrument switches to adiabatic mode and monitors the reaction through to completion. Tests are conducted in a small metallic sample bomb (approximately 10 ml, filled with 1–6 g of sample) equipped with a pressure transducer and temperature sensor.
ARC has a phi factor typically ranging from 1.5 to 3, meaning mathematical correction of the raw data is necessary before direct application to plant scale. The onset temperature, if left uncorrected, requires a safety margin of 20–40°C depending on the application.
From ARC testing, you can obtain:
- Adiabatic temperature rise and reaction heat
- Temperature and pressure rise rates
- Onset temperature of exothermic reactions
- Activation energy, reaction order, and pre-exponential factor
- Time to maximum rate (TMR) and TD24
- Temperature of no return (TNR)
- Self-accelerating decomposition temperature (SADT)
- Vapor-liquid phase equilibrium data
- Identification of autocatalytic behavior
The applicable testing standard is ASTM E1981 — Standard Guide for Assessing Thermal Stability by Accelerating Rate Calorimetry.
Reaction Calorimetry
While DSC and ARC characterize how a material behaves in isolation, reaction calorimetry measures the heat generated or absorbed during an actual chemical reaction under simulated process conditions. Reaction calorimeters replicate key features of stirred tank reactors — including dosing, mixing, and temperature control — making them uniquely suited for studying reaction pathways, assessing process conditions, and capturing thermal changes in real time as a reaction progresses.
In process safety, this makes reaction calorimetry especially important for scale-up work. Data on heat flow, energy output, gas evolution, and unreacted material accumulation directly informs heat removal requirements, reactor design, and process control strategies — reducing the risk of thermal runaway when transitioning from lab to pilot or full-scale production.
From reaction calorimetry testing, you can obtain:
- Heat flow and total energy output
- Gas evolution rates
- Unreacted material accumulation (dosing studies)
- Heat removal requirements for process control
- Data supporting safe scale-up and reactor design
There is no single global standard for reaction calorimetry, but industry guidance including CCPS Guidelines for Chemical Reactivity Evaluation and ASTM E1981 provide widely accepted frameworks. Reaction calorimetry is often used in combination with other hazard assessment methods to build a complete safety picture.
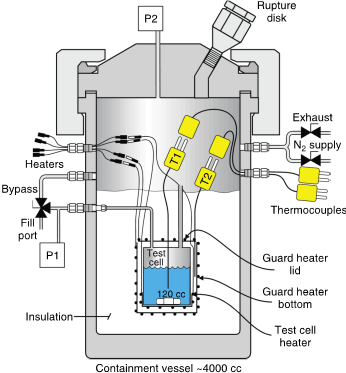
Vent Sizing Package (VSP)
The VSP was developed as part of the DIERS project in the 1980s specifically to address a limitation common to other calorimeters — the overestimation of thermal inertia, which leads to underestimating self-heat rates and adiabatic temperature rise at plant scale. Its defining feature is a low phi factor (typically 1.05 to 1.15), achieved by using a larger volume of reactants (~80 ml) in a thin-walled metal test cell. This allows laboratory-scale runaway reactions to accurately mirror the severity of those occurring in full-scale industrial vessels.
To compensate for the thin-walled cell’s limited pressure resistance, the test cell is placed inside a pressurized autoclave with a pressure controller that maintains a controlled pressure difference through nitrogen injection. A guard heater maintains adiabatic conditions, and a magnetic agitator allows reactions to be conducted under mixing conditions that closely resemble industrial processes.
VSP testing can be configured in several ways depending on the information needed:
- Closed cell test
- Open cell test for gas generation rate
- Open cell tempering test
- Blowdown top-vent test for vapor-liquid disengagement characterization
- Blowdown bottom-vent test for viscosity characterization
From VSP testing, you can obtain:
- Adiabatic temperature rise and reaction heat
- Temperature and pressure rise rates
- Onset temperature of exothermic reactions
- Gas generation rate and tempering capability
- Identification of flow regime during vessel venting (foamy vs. non-foamy)
- Viscosity data and its effect on relief flow capacity
- Time to maximum rate (TMR) and TD24
- Temperature of no return (TNR)
- Self-accelerating decomposition temperature (SADT)
- Required data for emergency relief system sizing
VSP testing aligns with API 520 and API 521 for pressure relief system design, and supports compliance with DIERS technology guidelines.
How These Methods Work Together
Reactive chemical testing rarely involves a single method in isolation. In practice, assessments typically follow a progression from screening to detailed characterization, with each stage informing the next.
DSC is almost always the starting point. Its speed, small sample requirement, and broad sensitivity make it ideal for initial screening — flagging whether a material has any thermal hazards worth investigating further. DTA and TGA are often run alongside DSC to fill in specific gaps, such as identifying gas evolution, mass loss, or thermal transitions that DSC alone may not fully characterize.
If DSC results indicate significant exothermic activity or runaway potential, ARC is the natural next step. It provides the adiabatic data needed to understand how a reaction would behave without heat dissipation — the worst-case industrial scenario. For reactive processes rather than pure materials, Reaction Calorimetry takes a parallel role, capturing heat flow and energy data under actual process conditions to support scale-up decisions.
VSP comes into play when emergency relief system design is required. It takes the understanding built through earlier testing and generates the specific pressure and temperature data needed to size relief devices accurately.
Not every assessment requires all six methods. The right combination depends on the material, the process, and the decisions that need to be made — but understanding how the methods relate to each other helps ensure that testing is targeted rather than redundant.