We also offer
Prime Process Safety Center performs Differential Scanning Calorimetry (DSC) testing on reactive chemicals, energetic materials, and unstable substances to identify thermal hazards before they reach your process. Using ASTM E537, our ISO/IEC 17025:2017 accredited lab measures heat flow events like decomposition onset, exothermic energy release, and phase transitions with small samples as little as 5–20 mg. Results are delivered in a written technical report with data interpretation and tailored recommendations.
What our Differential Scanning Calorimetry (DSC) test delivers
A single Differential Scanning Calorimetry (DSC) test on your material generates the following data:
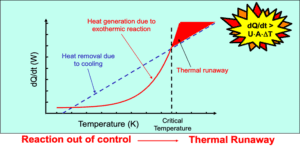
- Onset temperature of decomposition — the temperature at which your material begins to release energy. This establishes the upper boundary for safe processing and storage temperatures.
- Heat of decomposition (ΔH) — the total energy released during a thermal event, reported in J/g. Values exceeding 800 J/g indicate a potentially explosive material requiring further evaluation.
- Exothermic and endothermic event profiles — the full thermal curve showing melting points, crystallization, phase transitions, and reaction behavior across the test temperature range.
- Thermal stability classification — whether your material poses a low, moderate, or high thermal hazard based on ASTM E537 criteria.
Where DSC results indicate significant exothermic activity, we recommend follow-up testing with Accelerating Rate Calorimetry (ARC) to characterize self-heating behavior under adiabatic conditions to provide the additional data needed for relief system design and safe scale-up.
When to request Differential Scanning Calorimetry (DSC) testing
DSC testing is the appropriate first step when your material or process meets any of the following conditions:
- You are working with a new or unfamiliar chemical and need to establish its thermal stability before scale-up or production.
- Your process involves elevated temperatures and you need to confirm that operating conditions remain below the onset of any exothermic decomposition.
- Your material is being considered for transport or storage and regulatory or safety requirements call for documented thermal hazard data.
- You are screening a batch of materials for chemical incompatibilities and need a fast, small-sample method before committing to more resource-intensive testing.
- Your Safety Data Sheet (SDS) lacks thermal stability data and you need test-backed values to meet OSHA PSM or EPA RMP requirements.
- You need a cost-effective screening test before investing in higher-tier calorimetry such as ARC or Reaction Calorimetry.
DSC is particularly well suited for early-stage hazard screening because it covers a wide temperature range in a single run and requires minimal sample quantity.
How the Differential Scanning Calorimetry (DSC) test works
Principle of Operation
Differential Scanning Calorimetry compares the heat flow of your test sample against a reference material under controlled temperature conditions. Both pans follow the same heating program, and the instrument continuously monitors heat flow differences. The resulting thermal profile reveals events such as melting points, exothermic reactions, or decomposition behavior.
Sample and Cell Preparation
A small sample of 5–20 mg is placed in a test cell, typically stainless steel, aluminum, or gold-plated. For process safety studies, sealed high-pressure cells are recommended to prevent evaporative losses and better reflect real process conditions.
Temperature Program and Measurements
The DSC ramps the sample and reference pan at a controlled rate of 1–20 K·min⁻¹. Higher heating rates may reduce sensitivity in detecting onset temperatures, so rate selection is matched to the nature of your material and the purpose of the test. Throughout the run, the instrument measures exothermic and endothermic reactions, quantifies total heat release or absorption, and captures data on reaction energetics. Isothermal tests allow study of autocatalytic reactions, while varying ramp rates can yield kinetic data.
Data Interpretation
Results are displayed as a power versus time graph. Our engineers calculate reaction onset temperatures and energy release in J·g⁻¹, with safety factors applied. At higher heating rates above 5 K·min⁻¹, onset temperatures can appear artificially elevated, so rate selection and safety factor application are handled carefully. Comparing tests run under air and nitrogen atmospheres distinguishes oxidative reactions from pure decomposition. Where needed, multiple ramp rate tests allow extraction of kinetic parameters including activation energy, reaction order, and pre-exponential factor.
Key Results Your Report Will Include
- Reaction heat (J·g⁻¹)
- Onset temperature of exothermic and endothermic events, with safety factors applied
- Kinetic parameters where applicable (activation energy, reaction order, pre-exponential factor)
- Induction time for autocatalytic reactions
- Atmospheric comparison data where air and nitrogen runs are performed
- Flagging of decomposition energy values that may indicate explosive potential
Why Choose Prime Process Safety Center
Process Safety Is Our Only Focus
Unlike general analytical labs that offer DSC as one of dozens of services, Prime Process Safety Center specializes exclusively in process safety. Every test we run, including DSC, is interpreted through the lens of real industrial risk — not just material characterization.
Engineers, Not Just Technicians
Our team includes process safety engineers who understand how DSC findings connect to your broader operation. When results raise questions about scale-up risk, safe operating limits, or the need for further testing, you get engineering judgment alongside the data — not just a number on a page.
Full Reactive Hazard Testing Capability
When DSC results indicate the need for further evaluation, we can take that work forward in-house. Our capabilities include Differential Thermal Analysis (DTA), Thermogravimetric Analysis (TGA), Accelerating Rate Calorimetry (ARC), Reaction Calorimetry, and Vent Sizing Package (VSP) testing — so your hazard assessment stays with one team from screening through to design-level data.
FAQ
1. What is a Differential Scanning Calorimeter (DSC)?
A Differential Scanning Calorimeter (DSC) is an analytical instrument used to measure the heat flows associated with temperature changes in a sample. It provides information on phase transitions, thermal behavior, and energy changes in materials, making it valuable in process safety evaluations.
2. How does DSC work?
In DSC, the heat flow difference (or differential heat flow) between a sample and a reference material is measured as they undergo a controlled heating or cooling program. The energy differences indicate phase transitions, reactions, and other thermal events occurring in the sample. DSC data contributes to understanding the thermal behavior and safety aspects of materials.
3. What are the applications of DSC in process safety?
DSC has diverse applications in process safety assessments, including:
- Determining thermal stability and decomposition behavior of materials under process conditions.
- Identifying potential hazards associated with exothermic reactions, phase changes, or heat flow abnormalities.
- Assessing the compatibility of materials in applications involving temperature variations.
- Evaluating the influence of additives or impurities on the thermal behavior and reactivity of substances.
- Validating stability of active pharmaceutical ingredients (APIs) and other chemical compounds.
4. What information can be obtained from DSC measurements?
DSC measurements provide valuable insight into the thermal properties of materials, including:
- Detection and characterization of phase transitions such as melting points, crystallization, glass transitions, and more.
- Determination of reaction enthalpies and kinetic parameters such as activation energies.
- Evaluation of specific heat capacity and thermal conductivity.
- Quantification of heat flow changes associated with physical or chemical processes.
5. Can DSC data be used for process optimization?
While DSC primarily focuses on characterizing the thermal behavior and potential hazards of materials, the information obtained from DSC experiments can be applied to process optimization. By understanding the temperature conditions at which materials undergo phase transitions or reactions, process parameters can be optimized to minimize risks, enhance product quality, and improve overall process efficiency.