We also offer
Prime Process Safety Center performs Accelerating Rate Calorimetry (ARC) testing on reactive chemicals, energetic materials, and unstable mixtures to quantify thermal runaway behavior under adiabatic conditions. Using ASTM E1981 in our ISO/IEC 17025:2017 accredited lab, ARC testing determines the onset temperature of exothermic activity, the rate at which heat and pressure develop, and the kinetic parameters needed to establish safe operating limits, storage conditions, and transportation classifications. Results are delivered in a written technical report with full data interpretation and safety-specific recommendations.
What our Accelerating Rate Calorimetry (ARC) test delivers
A single ARC test generates the following data on your material’s thermal runaway behavior:
Onset temperature of exothermic reactions — the lowest temperature at which self-heating is detected above the instrument threshold, reported with a recommended 20–40°C safety margin depending on application.
Adiabatic temperature rise and reaction heat — the total temperature increase and energy released if the reaction proceeds without heat loss, directly informing worst-case consequence modeling.
Temperature and pressure rise rates — how quickly heat and pressure develop once exothermic activity begins, providing the quantitative basis for emergency relief system design and reactor safeguarding.
Time to Maximum Rate (TMR) and TD24 — the time available before a runaway reaction reaches its peak rate, and the temperature at which TMR equals 24 hours, both critical for establishing safe storage and handling conditions.
Self-Accelerating Decomposition Temperature (SADT) and Temperature of No Return (TNR) — key parameters for transportation classification and the identification of conditions beyond which spontaneous runaway cannot be prevented.
Reaction kinetic parameters — activation energy, reaction order, and pre-exponential factor, enabling predictive modeling of thermal behavior across different scales and conditions.
Detection of autocatalytic behavior — identification of reactions that accelerate due to their own products, including induction time quantification where applicable.
Vapor-liquid phase equilibrium data and gas generation — pressure profile analysis distinguishing vapor pressure contributions from permanent gas generation during decomposition.
When to request Accelerating Rate Calorimetry (ARC) testing
ARC testing is typically requested when a material or process requires more detailed thermal hazard data than screening methods such as DSC can provide. It is the appropriate next step when:
- Your DSC or DTA results show significant exothermic activity and you need accurate onset temperatures, runaway kinetics, and pressure data under adiabatic conditions.
- You are scaling up a chemical process from laboratory to pilot or industrial scale and need worst-case thermal data to design appropriate safeguards.
- Your process involves reactive intermediates, unstable compounds, or energetic materials that may decompose during heating, storage, or transportation.
- You need to establish SADT or TMR values for transportation classification or regulatory documentation under DOT, ADR, or UN requirements.
- You are investigating an abnormal temperature or pressure event during processing and need adiabatic data to understand how quickly conditions could escalate.
- You are designing or validating emergency relief systems and need accurate heat and pressure rise rates as inputs.
- Your material is being evaluated for long-term storage and you need to determine the temperature at which self-heating becomes a risk over time.
How the Accelerating Rate Calorimetry (ARC) test works
Principle of Operation
The ARC operates in two primary modes:
-
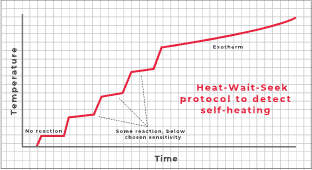
Heat-Wait-Search (HWS) mode
-
Isothermal Age mode
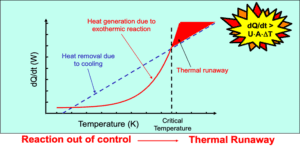
The key principle is to keep the sample under adiabatic conditions once exothermic activity or self-heating is detected. Under these conditions, the reaction accelerates due to the heat generated.
Although ARC experiments can be time-consuming, they are highly effective in capturing reactions with low self-heating rates that could otherwise go unnoticed.
Test Method
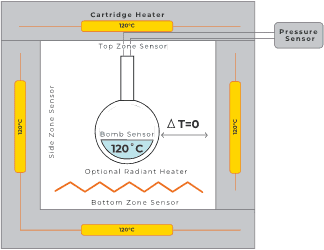
The test is performed using a 10 ml metallic sample bomb filled with 1 to 6 g of material. Depending on the chemical properties, spherical bombs made from different metals are used to avoid corrosion or catalytic interference.
The bomb, fitted with a pressure transducer and temperature sensor, is placed inside an adiabatic enclosure.
-
In the HWS method, the sample is step-heated with a radiant heater.
-
After each step, the system equilibrates and checks for exothermic activity above the threshold (typically 0.02 K·min⁻¹).
-
Once activity is detected, the system continuously monitors the reaction under strict adiabatic conditions until completion.
The ARC cell typically has high thermal inertia (phi factor 1.5–3), excellent pressure capacity (up to several hundred bar), and high sensitivity. However, since the vessel is usually not agitated, studying biphasic systems or triggering reactions with remote additions can be challenging.
Data Interpretation
The onset temperature of an exotherm is the lowest temperature where the calorimeter detects heat release above its set threshold. Analyzing the raw temperature, pressure, and time data provides key insights:
-
Reaction rates and magnitudes
-
Heat of reaction, calculated if thermal inertia and sample heat capacity are known
-
Kinetic parameters, including activation energy
Even though the ARC has a relatively high thermal inertia, results can be corrected using the phi factor. This correction helps determine parameters such as:
-
Time to Maximum Rate (TMR)
-
Self-Accelerating Decomposition Temperature (SADT)
For safety, the onset temperature should be applied with a 20–40°C margin, depending on application.
Why Choose Prime Process Safety Center
Process Safety Is Our Only Focus
Unlike general analytical labs that offer ARC as one of many services, Prime Process Safety Center specializes exclusively in process safety. Every ARC test we run is interpreted in the context of real industrial risk — connecting onset temperatures, runaway kinetics, and pressure data directly to the process conditions and safeguarding decisions your facility faces.
Engineers, Not Just Technicians
Our team includes process safety engineers who understand how ARC findings feed into downstream decisions — whether that’s emergency relief system design, transportation classification, or determining whether a process can be safely scaled. You get engineering judgment alongside the data, not just a report.
Full Reactive Hazard Testing Capability
ARC is most valuable when placed in the context of a complete hazard assessment. When DSC or DTA screening identifies exothermic activity that warrants further evaluation, we can progress directly to ARC testing in-house. Our capabilities also include Thermogravimetric Analysis (TGA), Reaction Calorimetry, and Vent Sizing Package (VSP) testing — so your assessment stays with one team from initial screening through to design-level data.
ISO/IEC 17025:2017 Accredited Laboratory
ARC testing at Prime Process Safety Center is performed in our ISO/IEC 17025:2017 accredited laboratory. Accreditation means our testing procedures, equipment calibration, and data quality are independently verified — giving you confidence in the results and the documentation that supports regulatory submissions, safety reviews, and insurance requirements.
FAQ
1. What is an Accelerating Rate Calorimeter (ARC)?
An Accelerating Rate Calorimeter (ARC) is a specialized instrument used to evaluate the thermal hazards and determine the temperature and pressure rise rate of reactions, mixtures, and materials. It is extensively used in process safety assessments to identify hazards related to exothermic reactions and reactive materials.
2. How does an ARC work?
An ARC typically consists of a small 10 ml sample bomb that holds the reaction or mixture being analyzed. The instrument monitors the temperature of the sample while a controlled heat source is applied, allowing for the measurement and assessment of the heat release rate. The ARC provides valuable data on the potential dangers of a reaction, including onset temperature, self-heating rates, heat of reaction, and time to maximum rate of heat release.
3. Why is an ARC important in process safety?
ARC plays a critical role in process safety by providing insights into the thermal behavior and hazards of reactions. It helps identify potential runaway reactions and thermal explosions, aids in designing safe reaction conditions, and assists in evaluating and selecting appropriate preventive measures for process safety.
4. What are the advantages of using an ARC?
Using an ARC offers several advantages, such as:
- Early identification of potential thermal hazards during a reaction or with reactive materials.
- Quantitative measurement of heat release rates, allowing for the accurate assessment of reaction hazards.
- Generating valuable data for process safety assessments and enabling the development of effective safety protocols.
- Assisting in the selection and evaluation of preventative measures, such as temperature control, venting systems, or the use of specialized equipment and materials.
5. Can an ARC be used for scale-up considerations?
While an ARC primarily focuses on evaluating small-scale thermal hazards, the data obtained from its measurements can be informative for scale-up considerations. By providing insights into the heat release rate and potential thermal hazards, an ARC can help guide the design and safety considerations when scaling up a process. However, additional measurements and analyses would usually be required to ensure safety at larger production scales.