We also offer
What is Differential Thermal Analysis (DTA)
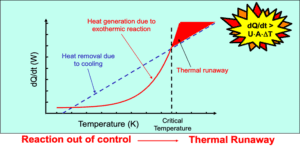
Monitoring the exothermic behavior of materials using Differential Thermal Analysis (DTA) is critical for identifying potential runaway reactions and hazardous conditions in a process. Sudden or significant heat releases can cause temperature spikes that exceed equipment control capabilities, potentially leading to thermal runaway, explosions, or other dangerous scenarios.
In process safety, common DTA lab equipment includes the Carius tube and the Rapid Screening Device (RSD). These tools are especially valuable because they allow simultaneous measurement of temperature and pressure, helping you understand material behavior under reactive conditions.
Why Perform Differential Thermal Analysis (DTA) Test?
Differential Thermal Analysis (DTA) testing is performed to better understand how materials behave when exposed to changing temperatures during storage, processing, or transportation. These insights help engineers anticipate thermal behavior that could affect safety, product quality, or process reliability.
DTA testing is commonly used to:
-
Screen materials for unexpected thermal activity during heating or cooling.
-
Assess how temperature changes influence process materials before scaling up production.
-
Compare thermal responses between raw materials, intermediates, or final products.
-
Investigate temperature-related issues encountered during manufacturing or handling.
-
Provide supporting data for process development, safety evaluations, and quality control.
By revealing how a material responds to temperature changes, DTA testing helps engineers make informed decisions about process conditions, material selection, and operational safeguards.
When Is Differential Thermal Analysis (DTA) Testing Needed?
Differential Thermal Analysis (DTA) testing is typically performed when temperature-related behavior of a material needs to be better understood before it is processed, stored, or used in a chemical operation.
DTA testing may be needed when:
-
Evaluating a new material or formulation before introducing it into a process.
-
Investigating unexpected thermal behavior observed during production or laboratory work.
-
Comparing thermal responses between raw materials, intermediates, or product batches.
-
Assessing materials exposed to elevated temperatures during drying, heating, or reaction steps.
-
Supporting process development or scale-up, where temperature effects may change under larger operating conditions.
-
Providing thermal analysis data for safety documentation, regulatory reviews, or technical reports.
Principle of Operation
Differential Thermal Analysis (DTA) works by heating a small sample while measuring the temperature difference between the sample and an inert reference material. Heating typically occurs at a constant rate. Exothermic or endothermic reactions within the sample release or absorb heat, which is recorded as deviations in the temperature difference by a differential thermocouple.
This test provides a semi-quantitative assessment of thermal characteristics, including exothermic, endothermic, and gas generation events. DTA can be applied to liquids, solids, or mixtures, making it versatile for many process safety applications.
Test Method
In an RSD or Carius tube test:
-
Place a small amount of material (1–6 mL in RSD, 10–15 mL in Carius tube) inside the test cell.
-
Position the cell in the center of a furnace and connect it to a pressure transducer.
-
Gradually increase the temperature at a fixed rate (e.g., 0.5 K/min) until either 400°C or the cell pressure limit is reached.
-
Observe deviations from the baseline temperature difference to detect exothermic or endothermic reactions.
Reactive substances may be introduced, and stirring can be applied to monitor reaction behavior accurately.
Data Interpretation
DTA results are presented in three key graphs:
-
Temperature, Pressure, and Time Trace – Shows the complete profile of the experiment.
-
Temperature Difference vs. Temperature – Highlights deviations indicating thermal events.
-
Natural Log of Pressure vs. Reciprocal of Absolute Temperature (Antoine Plot) – Reveals gas generation onset and rate.
-
The onset temperature is recorded at the first deviation, with upward shifts indicating exothermic reactions and downward shifts showing endothermic reactions.
-
A safety factor of ~50 K is applied to account for heat losses.
-
Peak height and width provide qualitative insights; broader peaks suggest mass transfer–controlled reactions.
-
Residual tube pressure after cooling quantifies gases released. Mass spectrometry can determine gas composition.
From these analyses, you can obtain:
-
Vapor-liquid phase equilibrium data (Antoine plot)
-
Total gas generation quantification
-
Onset temperature of reactions (with safety factor)
-
Induction time of autocatalytic reactions (requires expert guidance)
Why Choose Prime Process Safety Center
At Prime Process Safety Center, we provide:
-
Expertise: Our team has extensive experience performing DTA for reliable and accurate results.
-
Quality: Strict testing protocols and quality controls ensure consistent outcomes.
-
Insight: We offer detailed data interpretation and recommendations for your specific materials or processes.
-
Support: Prompt and efficient service to help you make informed safety decisions.
FAQ
1. What is Differential Thermal Analysis (DTA)?
Differential Thermal Analysis (DTA) is a technique used to measure the temperature difference between a sample and a reference material as they are heated or cooled. It provides information on the phase transitions, melting points, reactions, and thermal behavior of substances, making it useful in process safety evaluations.
2. How does DTA work?
In DTA, the temperature of the sample and a reference material are simultaneously measured as they undergo a controlled heating or cooling program. A temperature difference between the sample and the reference material indicates thermal events, such as phase transitions or reactions. DTA data provides insights into the heat flow associated with these events.
3. What are the applications of DTA in process safety?
DTA finds applications in process safety assessments by helping identify potential hazards related to thermal events in materials such as phase transitions, decomposition, or reaction-induced exothermic events. It aids in understanding the thermal behavior of materials and assists in designing safe operating conditions and selecting suitable mitigation measures.
4. What information can be obtained from DTA measurements?
DTA measurements can provide valuable information, including:
- Identification of phase transitions, such as melting points, crystallization, and glass transitions, which are important in understanding material behavior and stability.
- Detection of exothermic or endothermic reactions occurring during the temperature ramp, indicating potential hazards.
- Quantification of gas generation.
- Determination of thermal stability and decomposition temperature ranges.
5. Can DTA data be used for process optimization?
DTA data primarily focuses on understanding the thermal behavior and potential hazards of materials. However, the information obtained from DTA experiments, such as melting points or reaction temperatures, can be crucial in optimizing process conditions. By designing processes to operate below certain critical temperatures or avoiding conditions that induce reactions or phase changes, process safety can be improved.